
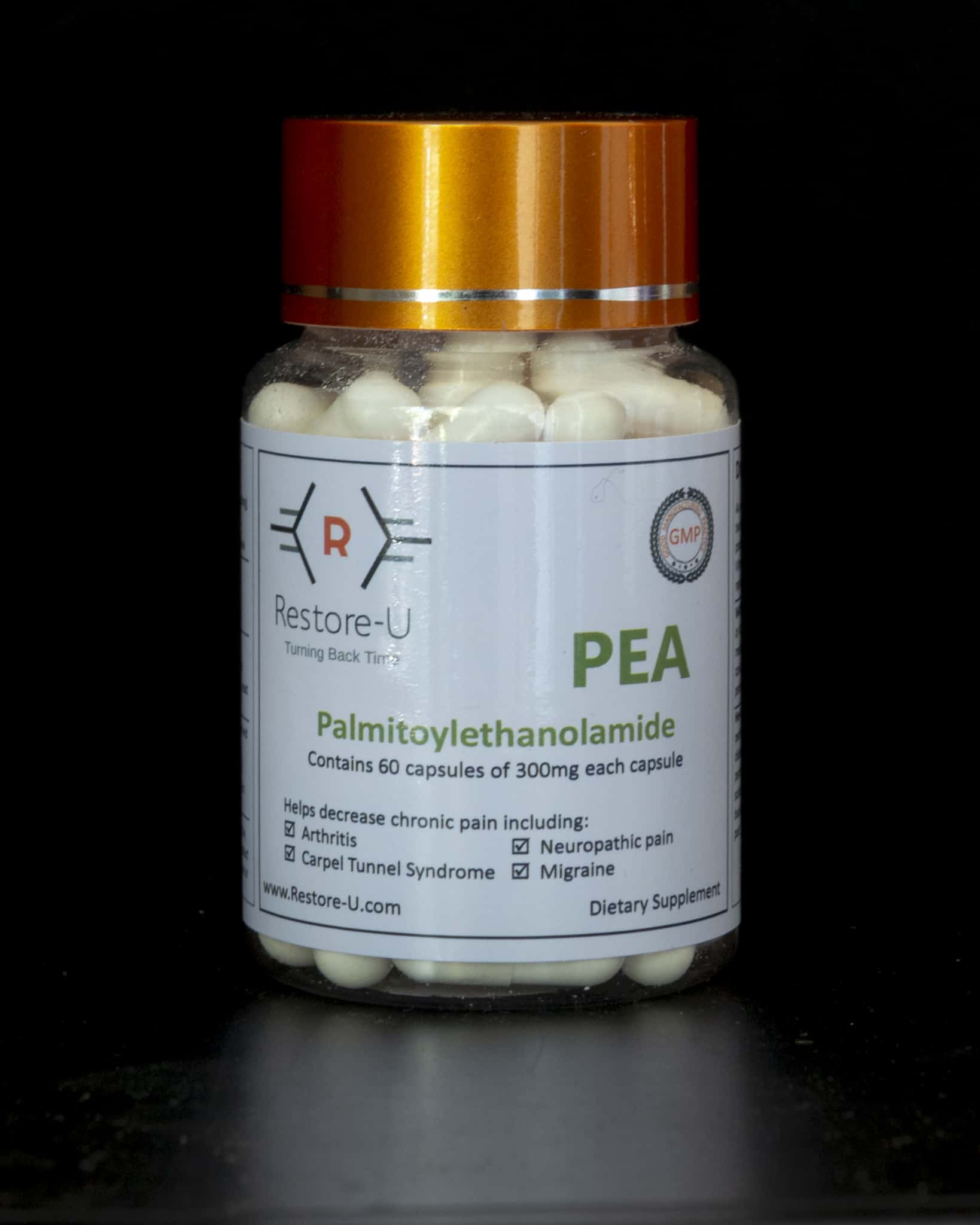
In 1957, Nobel Prize winner, Levi-Montalcini identified PEA as a naturally occurring molecule, describing its value in treating chronic infections and pains. Since her discovery, hundreds of scientific studies have been completed and have shown PEA is very effective and safe to use. PEA is described in scientific writings as a natural painkiller.
Indications for its use include:
Since 2012 a number of new trials have been published, among which studies in glaucoma.[48][49] PEA also seems to be one of the factors responsible for the decrease in pain sensitivity during and after sport, comparable to the endogenous opiates (endorphines).[50]
From a clinical perspective the most important and promising indications for PEA are linked to neuropathic and chronic pain states, such as diabetic neuropathic pain, sciatic pain, CRPS, pelvic pain and entrapment neuropathic painstates.[38][42][44][45][51][52]
In a blind trial, patients affected by pain from synovitis or TMJ osteoarthritis in total were randomly assigned to PEA or ibuprofen groups for two weeks. The decrease in pain reported after two weeks was significantly higher for the PEA-treated group, likewise for improved masticatory function.[53][54]
In 2012, 20 patients suffering from thalidomide and bortezomib induced neuropathy were reported to have improved nerve functions and less pain after a two-month treatment with PEA.[55] The authors pointed out that although a placebo effect might play a role in the reported pain relief, the changes in neurophysiological measures clearly indicated that PEA exerted a positive action on the myelinated fibre groups.
Sixteen men and fourteen women suffering from two major types of neuropathic pain refractory to analgesic treatment—peripheral diabetic neuropathy (4 men, 7 women) or post-herpetic neuralgia (12 men, 7 women)[56]—whose symptoms spanned eight pain categories ("burning", "osteoarticular", "piercing", etc.[57]) who were under prior treatment with pregabalin were transferred to PEA, after which pregabalin treatment was gradually reintroduced; all were responding well after 45 days, and presented significant decreases in painscores (without drug-drug interactions). [58]
In 2013, a metareview was published on the clinical efficacy and safety of PEA in the treatment of the common cold and influenza, based on reports from six double-blind, placebo, randomized controlled trials, addressing PEA's proposed anti-inflammatory and retinoprotectant effects.[4]
In 2016, titled: Palmitoylethanolamide for the treatment of pain: pharmacokinetics, safety and efficacy, found 21 clinical studies, of which 16 were clinical trials enrolling a range of 20 to 636 patients and five were case/pilot studies. In the clinical trials, PEA was used for periods ranging from 14 days to 120 days, and the doses ranged from 300 mg to 1200 mg daily. The authors concluded on the basis of their analyses that PEA was an effective treatment for pain with no registered serious adverse effects.
Findings include:-
Best results are achieved when taken, orally, for a period of 2 to 3 months with 300mg/1 capsule 2-3 times a day is the proven dose to start on.
There are no known problematic side effects. PEA can be taken together with any other substances without any side effects. It enhances the pain-relieving effect of classic analgesics and anti-inflammatories.
Anyone with a painful or chronic health disorder, even if you are taking medication for these complaints, as it has been shown to enhance the effect of medication. People who react to or find no benefit with anti-neuropathic or anti-convulsant drugs for nerve pain may benefit from PEA, this has been shown in large studies for sciatica and carpal tunnel syndrome.