Treating immune cells that target hepatitis B with nicotinamide mononucleotide (NMN) restores their DNA repair mechanisms and antiviral function.
By Brett J. Weiss https://www.nmn.com/news/new-european-study-restoring-nad-with-nmn-rescues-immune-cell-function-in-hepatitis-b-patients
Highlights
Hepatitis B typically resolves within six months for most people infected, but chronic hepatitis B that doesn’t go away can occur due to CD8 T cell exhaustion. CD8 T cell exhaustion results from high levels of exposure to a pathogen like the hepatitis B virus, triggering T cell DNA damage and a dysfunctional antiviral response. Identifying the cellular processes behind CD8 T cell exhaustion can help with developing therapies for chronic hepatitis B. Some researchers have recently proposed that NAD+ depletion underlies T cell exhaustion.
Published in the Journal of Hepatology, Fisicaro and colleagues from the University of Parma in Italy demonstrate that treating CD8 T cells specific for the hepatitis B virus with NMN improves their production of antiviral proteins – cytokines. Exhausted hepatitis B-specific CD8 T cells were found to have high levels of DNA damage along with dysfunctional DNA repair mechanisms. Findings from the study suggest that NMN and/or CD38 inhibitors can be used to revamp exhausted T cells to ward off chronic hepatitis B, along with potentially other viral infections.
Because Fisicaro and colleagues hypothesized that restoring NAD+ levels with NMN could revamp exhausted CD8 T cells, they isolated these immune cells from chronic hepatitis B patients for testing. Upon treatment with NMN, the cells expressed more antiviral cytokines, specifically a 2.7-fold increase for cytokine interferon gamma (IFN-𝛾), suggesting that NMN restores the CD8 T cells’ antiviral properties.
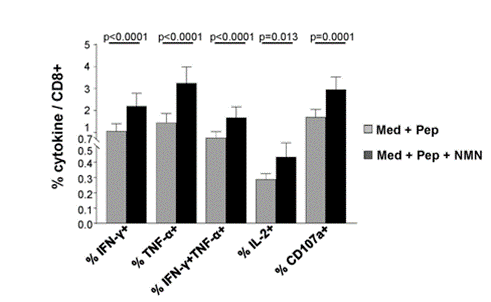
(Montali et al., 2023 | Journal of Hepatology) NMN restores immune cell antiviral cytokine production. Cellular cytokines listed on the X-axis significantly increased following immune cell treatment with NMN and hepatitis B viral proteins (black bars) compared to those treated with hepatitis B viral proteins alone (gray bars). Med + Pep= Cell culture medium with viral peptide stimulation; Med + Pep + NMN= Cell culture medium with viral peptide stimulation and NMN
To examine the cellular damage that goes along with T cell exhaustion in chronic hepatitis B infection, Fisicaro and colleagues compared DNA damage in hepatitis B-specific T cells with influenza (FLU)-specific T cells. The researchers found significantly more DNA damage in the hepatitis B-specific T cells.
They also found that treating the cells with a DNA damage-inducing molecule (etoposide) elicited a trend toward a lower DNA damage response compared to FLU-specific T cells. Since DNA damage responses like the one mediated by PARPs require NAD+, this data suggests that NAD+ deficiency is tied to higher levels of DNA damage in exhausted hepatitis B-specific CD8 T cells.
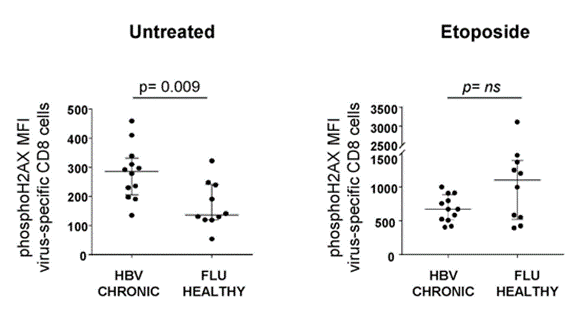
(Montali et al., 2023 | Journal of Hepatology) Hepatitis B-specific CD8 T cells have high levels of DNA damage and a weak DNA damage response to a DNA damaging molecule (etoposide). Left) Compared to FLU-specific CD8 T cells (FLU HEALTHY), those specific for hepatitis B (HBV CHRONIC) exhibit higher levels of markers for DNA damage (phosphoH2AX). Right) A molecule used to induce DNA damage (etoposide) elicits a trend toward lower DNA damage response in hepatitis B-specific immune cells (HBV CHRONIC) compared to FLU-specific immune cells (FLU HEALTHY), suggesting lower NAD+ required for DNA repair.
To explore further how NAD+ repletion with NMN improves exhausted CD8 T cell antiviral cytokine production, Fisicaro and colleagues measured how high levels of the NAD+-consuming CD38 enzyme relate to cytokine levels. They measured one of the primary cytokines – IFN-𝛾 – and found that high CD38 enzyme levels are associated with low IFN-𝛾 levels. These data provide evidence that high CD38 enzyme levels can lead to lower antiviral cytokine production. This also lends credulity to the assertion that low NAD+ levels play an important role in chronic hepatitis B pathology since high CD38 levels would theoretically lower cellular NAD+.
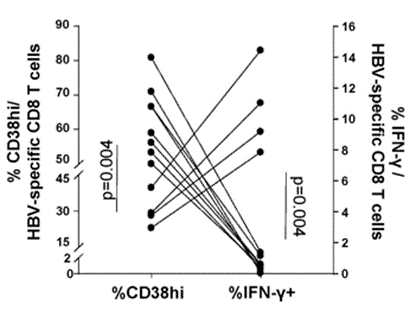
(Montali et al., 2023 | Journal of Hepatology) Higher immune cell CD38 enzyme levels are associated with lower antiviral cytokine levels. H igher percentages of immune cells with elevated CD38 (%CD38hi) are associated with a lower probability of expressing the antiviral cytokine IFN-𝛾 (%IFN-𝛾+).
“Our data show increased DNA damage with limited activation of the DNA repair machinery in [hepatitis B virus]-specific CD8 T cells from [chronic hepatitis B] patients,” say Fisicaro and colleagues. “This strongly suggests that NAD-consuming enzymes, particularly overexpressed CD38, may play a pivotal role in NAD depletion. Reconstitution of many interconnected intracellular functions by NMN supplementation indicates that NAD depletion likely represents an important determinant of T cell exhaustion.”
The study provides evidence that the hepatitis B virus triggers higher CD38 enzyme activation, leading to lower NAD+ levels. Since DNA repair mechanisms like PARPs require NAD+ for their function, this could help explain why exhausted hepatitis B virus-specific T cells have more DNA damage. This could also help explain why DNA repair mechanisms don’t work as well in exhausted hepatitis B-specific CD8 T cells.
Treating CD8 T immune cells with the NAD+ precursor NMN restores antiviral cytokine production, suggesting that NAD+ repletion reconstitutes their function. NMN’s capability to revamp immune cell function provides support that NAD+ depletion contributes to CD8 T cell exhaustion and dysfunction. Whether NMN provides beneficial effects in exhausted CD8 T cells in other types of infections should be explored. If NMN can restore exhausted T cells in other infections, it may be the case that NAD+ makes it easier for immune cells to fight other types of infection, also.
Model
Model: Hepatitis B-specific CD8 T cells from chronic hepatitis B patients
TAGS
Antiviral, Hepatitis, Immunity, Nicotinamide Mononucleotide, T cells
Story Source
Montali I, Berti CC, Morselli M, Acerbi G, Barili V, Pedrazzi G, Montanini B, Boni C, Alfieri A, Pesci M, Loglio A, Degasperi E, Borghi M, Perbellini R, Penna A, Laccabue D, Rossi M, Vecchi A, Tiezzi C, Reverberi V, Boarini C, Abbati G, Massari M, Lampertico P, Missale G, Ferrari C, Fisicaro P. Deregulated intracellular pathways define novel molecular targets for HBV-specific CD8 T cell reconstitution in chronic hepatitis B. J Hepatol. 2023 Mar 7:S0168-8278(23)00167-8. doi: 10.1016/j.jhep.2023.02.035. Epub ahead of print. PMID: 36893853.